Walking into a CBD shop in Miami, you face shelves packed with tinctures, gummies, and vapes, all promising wellness benefits. But here’s the unsettling truth: studies reveal frequent mislabeling, with products containing wildly different CBD levels than advertised, and some harboring dangerous contaminants like heavy metals or pesticides. For Miami consumers seeking genuine relief and safety, understanding purity and quality standards isn’t optional anymore. This guide cuts through the confusion, explaining exactly what testing methods reveal product safety, which contaminants to watch for, how regulations protect you, and practical steps to verify quality before you buy.
Table of Contents
- Key takeaways
- Methods for testing CBD purity and quality
- Key contaminants and purity benchmarks every Miami consumer should know
- Challenges in CBD labeling and regulatory landscape
- How to verify CBD purity and quality when shopping in Miami
- Shop quality CBD products with confidence locally
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Mislabeling risk | Studies show frequent mislabeling with CBD amounts that do not match the label and potential contaminants in some products. |
| Testing methods | Primary methods include liquid chromatography with photodiode array detection, high performance liquid chromatography with photodiode array detection, high performance thin layer chromatography, liquid chromatography mass spectrometry, and gas chromatography mass spectrometry to identify cannabinoids and detect impurities. |
| Best testing method | For the most accurate cannabinoid and contaminant profile, prefer products tested with liquid chromatography mass spectrometry. |
| Contaminants to watch | Key contaminants include heavy metals such as lead, cadmium, arsenic, and mercury; pesticides; residual solvents; microbes; and mycotoxins. |
| COA verification | Always check a third party Certificate of Analysis and understand that regulatory standards vary by region to inform safer purchases. |
Methods for testing CBD purity and quality
When you pick up a CBD product claiming 1000mg per bottle, how do you know that number reflects reality? The answer lies in sophisticated laboratory techniques that separate, identify, and quantify cannabinoids with precision. Primary methods for CBD purity testing include LC-PDA, HPLC-PDA, HPTLC, LC-MS/MS, and GC-MS for comprehensive cannabinoid profiling, potency verification, and impurity detection. These aren’t just fancy acronyms. They represent different approaches to analyzing what’s actually inside your wellness products.
Liquid chromatography paired with photodiode array detection (LC-PDA) and high-performance liquid chromatography (HPLC-PDA) work by dissolving CBD extracts in liquid solvents, then passing them through specialized columns that separate individual compounds based on their chemical properties. As each cannabinoid emerges, detectors measure light absorption patterns to identify and quantify substances like CBD, THC, CBG, and CBN. These methods excel at measuring cannabinoid profiling techniques for major and minor compounds without destroying samples.
Mass spectrometry techniques (LC-MS/MS and GC-MS) take analysis further by breaking molecules into charged fragments, then measuring their mass-to-charge ratios. This approach provides exceptional sensitivity and specificity, detecting trace contaminants and confirming molecular structures with near-certainty. Gas chromatography mass spectrometry (GC-MS) heats samples into vapor form before analysis, making it particularly effective for volatile compounds and residual solvents. Liquid chromatography mass spectrometry (LC-MS/MS) handles heat-sensitive cannabinoids without degradation, offering the most comprehensive profile available.
For Miami consumers evaluating product quality, understanding these methods matters because testing rigor directly impacts your safety. Cheaper, less precise methods might miss contaminants or misreport potency. Reputable manufacturers invest in multiple testing approaches to verify results across different analytical platforms. When you see a Certificate of Analysis listing the testing method, you gain insight into data reliability.
Pro Tip: Prefer products tested using LC-MS or LC-MS/MS methods for the highest accuracy and most comprehensive cannabinoid and contaminant profiles, especially when shopping through a trusted weed delivery marketplace that prioritizes transparency.
Key contaminants and purity benchmarks every Miami consumer should know
Purity testing reveals more than cannabinoid percentages. It exposes hidden dangers lurking in poorly manufactured products. Key contaminants include heavy metals like lead, cadmium, arsenic, and mercury, along with pesticides, residual solvents from extraction, microbes, and mycotoxins from mold contamination. Each category poses distinct health risks, particularly with regular consumption.
Heavy metals accumulate in your body over time, causing neurological damage, kidney problems, and increased cancer risk. Hemp plants naturally absorb metals from soil, making source material quality critical. Pesticides used during cultivation can survive extraction processes, introducing neurotoxins and hormone disruptors into final products. Residual solvents like butane, propane, or ethanol left over from extraction methods irritate lungs and organs when inhaled or ingested repeatedly. Microbial contamination from bacteria, yeast, or mold introduces pathogens that trigger infections, especially in immunocompromised individuals. Mycotoxins produced by certain molds damage liver function and suppress immune systems.

Regulatory bodies and industry standards establish safety thresholds for these contaminants. California’s Proposition 65 sets strict limits for heavy metals in consumer products. The United States Pharmacopeia (USP) provides reference standards for pharmaceutical-grade CBD. Purity benchmarks require CBD isolates exceed 97-99% purity with total unspecified impurities staying below 0.50%, while THC must remain at or below 0.3% dry weight to meet federal hemp definitions.
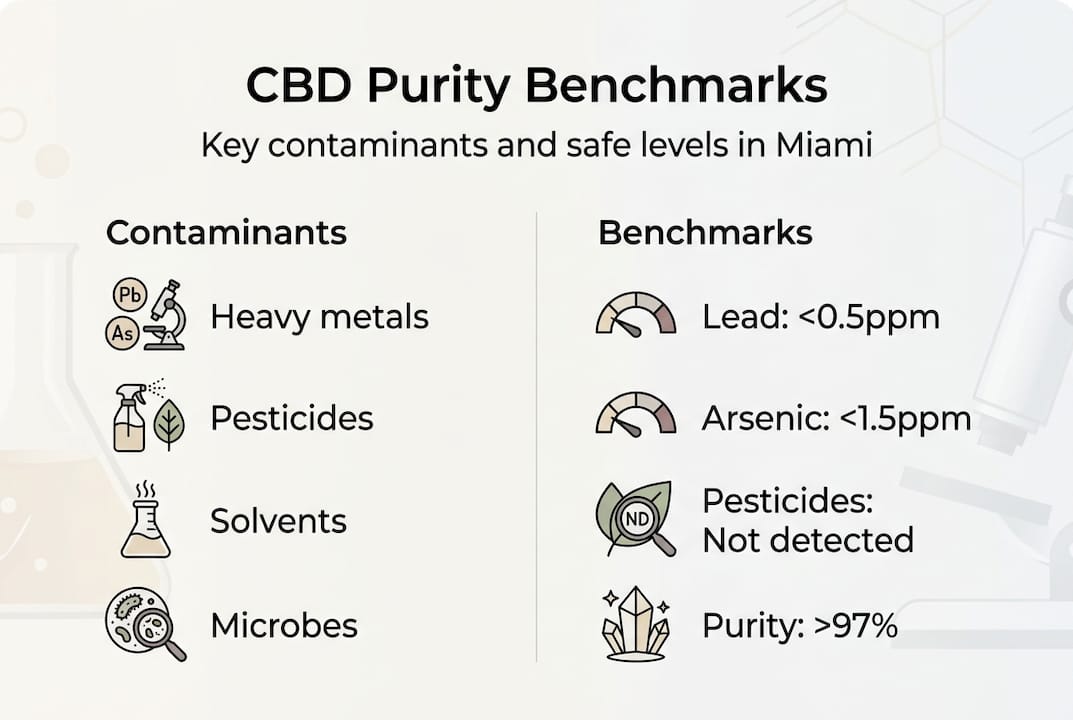
| Contaminant Type | Health Risks | Regulatory Threshold |
|---|---|---|
| Heavy Metals | Neurological damage, kidney failure, cancer risk | Lead <0.5 ppm, Cadmium <0.5 ppm, Arsenic <1.5 ppm, Mercury <3.0 ppm (USP limits) |
| Pesticides | Neurotoxicity, hormone disruption, developmental harm | Must pass state-specific pesticide panels (typically <0.1 ppm per compound) |
| Residual Solvents | Respiratory irritation, organ damage | Ethanol <5000 ppm, Butane <5000 ppm, Propane <5000 ppm (USP Class 3) |
| Microbes | Infections, immune suppression | Total aerobic count <10,000 CFU/g, yeast/mold <1,000 CFU/g |
| Mycotoxins | Liver damage, immunosuppression | Aflatoxins <20 ppb, Ochratoxin A <20 ppb |
Miami buyers should evaluate whether products meet these benchmarks before purchase. A CBD isolate claiming 99% purity with verified low impurity levels offers far greater safety than a full-spectrum oil with unknown contaminant status. Total impurities encompassing residual solvents, heavy metals, and other unwanted substances should never exceed half a percent in quality products.
Pro Tip: When reviewing lab reports for CBD product selection, verify that CBD isolate purity exceeds 97% and total impurities remain at or below 0.5% for optimal safety and effectiveness.
Challenges in CBD labeling and regulatory landscape
You might assume that products on store shelves accurately reflect their labels, but reality tells a different story. Empirical research demonstrates frequent mislabeling, with CBD content often understated or overstated by significant margins, and some third-party laboratories inflating potency results to satisfy clients. This systematic problem undermines consumer trust and creates genuine health risks when people dose based on inaccurate information.
Several factors drive labeling inconsistencies. Manufacturers sometimes use inferior testing methods or skip verification steps to reduce costs. Marketing pressures incentivize exaggerated potency claims to appear more competitive. Lack of standardized testing protocols across laboratories produces conflicting results for identical samples. Some companies deliberately mislabel products to circumvent regulations or mislead consumers about THC content. Without consistent oversight, these practices persist across the industry.
Regulatory frameworks governing CBD remain fragmented and evolving. Federal hemp regulations mandate Δ9-THC levels stay at or below 0.3% dry weight per the 2018 Farm Bill, defining the legal boundary between hemp and marijuana. However, states impose additional requirements that vary dramatically. New York limits total THC (including THCA) to 0.3%. Vermont requires CBD products undergo testing for potency and contaminants before sale. Colorado mandates specific label warnings and testing standards. Florida follows federal guidelines but adds its own hemp program requirements.
The Food and Drug Administration (FDA) continues wrestling with how to regulate CBD in foods, dietary supplements, and cosmetics. Currently, the FDA prohibits adding CBD to food products or marketing it as a dietary supplement without approval, yet enforcement remains inconsistent. This regulatory uncertainty leaves consumers navigating a marketplace where rules differ by jurisdiction and compliance varies by manufacturer. The USP advocates for uniform quality standards across the industry, but adoption remains voluntary for most producers.
For Miami consumers, these regulatory inconsistencies create practical challenges. Products legal in one state might violate rules in another. Lab reports from unaccredited facilities might contain errors or fabrications. Marketing claims often outpace scientific evidence. To navigate this landscape safely, you need to become an informed buyer who verifies claims independently rather than trusting labels at face value.
Take these actions to avoid mislabeled or unsafe products:
- Demand recent third-party lab reports (Certificates of Analysis) dated within the past six months for every product you consider
- Verify the testing laboratory holds ISO 17025 accreditation, confirming it meets international quality standards
- Cross-reference CBD and THC percentages between the label and lab report to catch discrepancies
- Check that contaminant screening includes heavy metals, pesticides, residual solvents, and microbial testing
- Research the brand’s reputation through independent reviews and consumer protection databases
- Avoid products making medical claims or promising to cure specific diseases, as these violate FDA regulations
- Purchase from retailers who provide transparent access to CBD regulations overview and testing documentation
How to verify CBD purity and quality when shopping in Miami
Armed with knowledge about testing methods, contaminants, and regulations, you can now take practical steps to verify product quality before spending money. The Certificate of Analysis (COA) serves as your primary verification tool, providing laboratory-confirmed data about what’s actually inside the bottle. Follow this systematic approach to evaluate any CBD product confidently:
-
Request the COA directly from the retailer or manufacturer before purchase, either in-store or through their website. Legitimate companies provide these documents readily and transparently. Hesitation or refusal signals potential quality issues.
-
Verify the testing laboratory’s credentials by checking for ISO 17025 accreditation, which confirms the facility meets international standards for testing competence and quality management. Look for the accreditation body’s logo and certificate number on the report.
-
Confirm the COA matches the specific product batch you’re buying by cross-referencing batch numbers, production dates, and product names. Generic or outdated reports don’t guarantee the quality of your particular purchase.
-
Examine the cannabinoid profile section for CBD percentage, THC levels, and other cannabinoids present. Quality products show <0.3% THC levels with CBD content matching label claims within a reasonable margin (typically ±10%).
-
Review the contaminant screening results for heavy metals, pesticides, residual solvents, and microbial counts. All values should fall below the regulatory thresholds outlined earlier, with clear “pass” designations for each category.
-
Check the testing methodology listed on the COA. Prefer reports showing LC-MS, LC-MS/MS, or HPLC methods for cannabinoid analysis, as these provide superior accuracy compared to less sophisticated techniques.
-
Note the report date to ensure freshness. CBD degrades over time when exposed to heat, light, and oxygen. Reports older than six months suggest the product has been sitting in inventory too long, potentially losing potency.
Beyond COA evaluation, watch for red flags that indicate quality problems. Synthetic delta-8-THC products often contain harmful isomerization byproducts and contaminants from chemical conversion processes. These semi-synthetic cannabinoids carry unknown health risks and often violate state regulations despite appearing in retail stores. Products stored in clear containers or exposed to bright lighting degrade faster, reducing CBD content and forming oxidation byproducts. Prices significantly below market average usually signal inferior source material, minimal testing, or diluted formulations.
Miami’s climate presents additional considerations. High heat and humidity accelerate CBD degradation, making proper storage critical. Choose products in dark glass bottles with airtight seals. Store them in cool, dark locations away from direct sunlight and heat sources. Refrigeration extends shelf life for oils and tinctures, though it may cause temporary cloudiness that disappears at room temperature.
When shopping from trusted CBD sources, verify they maintain proper storage conditions and rotate inventory regularly. Ask staff about product turnover rates and storage practices. Reputable retailers educate customers about quality indicators rather than pushing sales. They provide easy access to lab reports and answer questions about testing protocols transparently. Understanding CBD oil lab certificate details empowers you to make informed decisions based on objective data rather than marketing hype.
Pro Tip: Always verify the COA date falls within the past six months and confirms the test panel covers both cannabinoid potency and comprehensive contaminant screening to ensure you’re getting fresh, safe products.
Shop quality CBD products with confidence locally
Now that you understand purity standards, testing methods, and verification strategies, you can shop for CBD products with genuine confidence. Get Blazy CBD Dispensary serves Miami consumers who refuse to compromise on quality or transparency. Our Biscayne Boulevard location offers premium hemp-derived products from manufacturers who prioritize rigorous testing and accurate labeling. Every product in our inventory includes accessible lab reports showing cannabinoid profiles and contaminant screening results, so you verify quality before purchase.
Whether you prefer CBD oils, tinctures, gummies, vape products, or topical solutions, our knowledgeable staff helps you navigate options using the quality criteria outlined in this guide. We source from reputable manufacturers committed to purity benchmarks and regulatory compliance. Shop in-store for personalized guidance or order online for fast, discreet delivery throughout Miami. Visit our weed delivery marketplace to explore our full CBD product selection and experience the difference that transparency and quality standards make for your wellness journey.
Frequently asked questions
What are common contaminants in CBD products?
Common contaminants include heavy metals like lead, cadmium, arsenic, and mercury absorbed from soil during cultivation. Pesticides used on hemp plants, residual solvents from extraction processes, microbes from poor sanitation, and mycotoxins from mold growth also pose risks. Proper third-party testing ensures these harmful substances remain below established safety limits before products reach consumers.
How can I check if a CBD product is accurately labeled?
Request a recent Certificate of Analysis (COA) from an ISO 17025-accredited laboratory that tested the specific batch you’re purchasing. Compare the cannabinoid percentages on the lab report with label claims, checking that CBD content matches within reasonable margins and THC stays below 0.3%. Verify the report includes comprehensive contaminant screening with passing results for heavy metals, pesticides, solvents, and microbes.
Are all CBD products legal and safe in Miami?
Federally, hemp-derived CBD products must contain 0.3% or less delta-9-THC to remain legal under the 2018 Farm Bill. Miami follows these federal guidelines along with Florida’s hemp program requirements. However, legality doesn’t guarantee safety. Product safety depends entirely on manufacturing quality, purity standards, and proper testing. Always verify Certificates of Analysis before purchase to confirm both legal compliance and safety.
What should I look for on a lab report to ensure quality?
Look for a detailed cannabinoid profile showing CBD and THC percentages that match label claims, comprehensive contaminant screening confirming absence of heavy metals and pesticides below regulatory limits, and clear documentation of the testing laboratory’s ISO 17025 accreditation. Prefer reports using LC-MS or HPLC testing methods and dated within the past six months. The report should list specific values for each tested parameter rather than just pass/fail designations, giving you complete transparency about product composition and safety.